 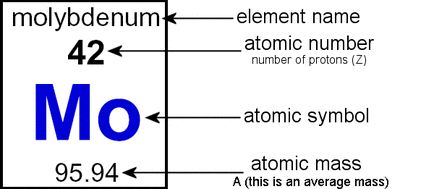
Difference between Mass Number and Atomic Mass. This concept was historically important because it provided a theoretical. Hence, for an atom mass number number of protons + number of neutrons. Carbon 12 will remain carbon 12 unless a fission or fusion reaction changes that. Atomic number is defined as the number of protons in the nucleus of an atom. Atomic numbers are always whole numbers greater than zero, such as 1,2. They all occur naturally in nature, but only carbon 12 is stable, and does not decay. An atomic number of an element is the number of protons in all the atoms of that element. For example, carbon 14, carbon 13, and carbon 12 atoms all have six protons, but they each have eight, seven, and six neutrons respectively. If you subtract the atomic number from the atomic mass, you will end up with the number of neutrons present in the atom, allowing you to determine the type of isotope it is. Atomic number definition, the number of positive charges or protons in the nucleus of an atom of a given element, and therefore also the number of electrons normally surrounding the nucleus. When you are looking at an isotope symbol, the atomic number is listed in the lower left hand corner of the symbol, while the atomic mass number is rounded to the nearest whole number and listed in the top left of the symbol. For ordinary nuclei, this is equal to the proton number (np) or the number of protons found in the nucleus of every atom of that element. 
The Periodic table of elements lists the atomic number above the element symbol. The atomic number or nuclear charge number (symbol Z) of a chemical element is the charge number of an atomic nucleus. The relationship between atomic number and isotopes is very important in the world of science. If a substance has 1 microgram of carbon 14 when it was created, it would only have 0.5 micrograms 6000 years later, 0.25 micrograms 12,000 years later and son on. For example, carbon 14 decays to carbon 12 at a rate of half of the carbon 14 isotopes per 6,000 years. The natural ratio of carbon 14, 13, and 12 is known, just as the half-life of these isotopes. This is a great example of how Chemistry, the central science, has made a groundbreaking contribution to other sciences (in this case geology). The relative amount of carbon isotopes in a substance is what allows scientists to perform radiocarbon dating (radioactive dating also works for other isotopes). September 3, Glasshouse- I would like to add to what you have said. No kidding, I think only 3 other people that I know use this website. Thank you WiseGeek! You have saved my science project! More people should know about this website. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
